Photos / Sounds
What
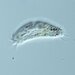
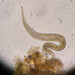
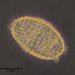
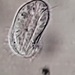
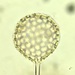
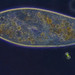
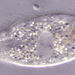
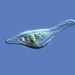
Genus CentropyxisObserver
pezzottal7Description
Water taken from the fountain in the third picture. Possibly C. discoides?
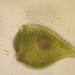
Photos / Sounds
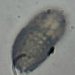
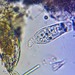
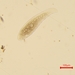
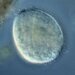
What
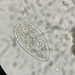
Lobose Amoebae (Subphylum Lobosa)Observer
hgmuellerDescription
Size about 120 um x 100 um
This has to be the slowest moving amoebae I've ever seen. I had passed by dozens of these thinking they were expired protists but when I finally focused on one and watched it I saw SLOW movement.
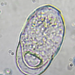
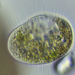
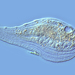
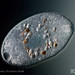
Photos / Sounds
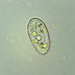
What
Caenomorpha capucinaObserver
peptolabDescription
Caenomorpha capucina Kahl, 1932 from the benthos of Georgica Cove, a brackish (5 ppt salinity) landward offshoot of estuary Georgica Pond. Imaged in Nomarski DIC on Olympus BH2S using SPlan 40 0.70 objective plus variable phone camera cropping on Samsung Galaxy S24. The cell body length is 80 um and total length including caudal spine is 100 um. The size (based on Kahl's illustration labelled 100 um), the quite coarse granulation, the configuration of the two spines, and the macronucleus shape (sausage-shaped with large round micronucleus), are diagnostic. Jankowski notes that macronucleus size and shape are quite strong characters to differentiate Caenomorpha species other than C. medusula (1).
The species is listed in Carey 1992: "Caenomorpha capucina Kahl, 1932 Helmet-shaped or medusoid, 50-70 µm in length. There are two posterior spines. The macronucleus is large and sausage-shaped". Kahl's original description: "Caenomorpha capucina Size of Body 50-70 um, caudal spines just as long, secondary spines little shorter. The frontal ridge forms a strongly pronounced ectoplasmic fold to the right. The front, as a narrow cap extends over and so also forms a clearly splayed edge running parallel to it in front of the ciliated bell edge. After overlap of the right, this edge converges on the dorsal side to the main seam, and runs into the base of the tail. Cytoplasm with coarse, dark appearing reserve bodies, which are particularly clustered at the front. Macronucleus pretty long sausage-shaped, micronucleus large, round, slightly stainable. The most complicated Caenomorpha; extremely interesting in terms of phylogenetic history as the final link in the series of developments: med., lauterborni; levanderi. Opposite of the strong development of the strongly spiral ridges with their three secondary seams the adoral zone is very reduced (less than the last half turn). On the first half are the rudimentary membranelles hidden in cilia. Not uncommon in the sapropel of the bridge bay of Schilksee (Kiel); the ventral hilltop is also very fond of stunted forms; is the secondary spine rarely rudimentary" (1).
The congener C. lauterborni is smaller at 70 um, has an ovoid macrunculeus (vs sausage shaped in C. capucina), has a spine 2 that is curved toward the much longer caudal spine 1 while in C. capucina the two spines are roughly parallel and closer in length. Jankowski notes that "spine 2 is poorly developed in a majority of C.lauterborni; it looks sometimes like a short needle-like somewhat curved prominence of an armour in the contractile vacuole area" (2). Another congener, C. levanderi, is reported as similar in size per Kahl (80-100 um) (1) or smaller (60-70 um per Jankowski) (2), has an elongated ovoid (2) or ellipsoid short sausage-shaped (1) macronucleus, and the spine 2 never develops here unlike C. lauterborni (2). Neither of these congeners show the coarse granular aggregate of C. capucina (1,2). . Congeners are depicted at the end of the illustrations.
- A. Kahl; [Urtiere oder Protozoa I: Wimpertiere oder Ciliata (Infusoria) 3. Spirotricha]. Die Tierwelt Deutschlands und der angrenzenden Meeresteile. Germany:Verlag von Gustav Fischer. (3), 1932
- Morphology and Evolution of Ciliophora. III. Diagnoses and Phylogenesis of 53 Sapropelebionts, Mainly of the order Heterotrichida By AXATOLY. JANKOWSKI Arch. Protistenk. Bd. 107,pp 185-294 (1961)
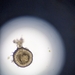
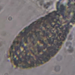
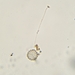
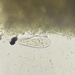
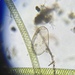
What
Trinema galeataObserver
nolan1737Description
I'm not sure what this could be, can you identify it?
Photos / Sounds
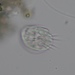
What
Aspidisca cicadaObserver
someplantDescription
Magnification of all photos: 400×
Habitat: fine particulate matter attached to rocks in a stagnant part of the River des Peres.
Using stage micrometer for calibration, cell body (excluding cirri) estimated to be about 37 µm long.
Photos / Sounds
What
Trochiliopsis opacaObserver
peptolabDescription
Trochiliopsis opaca Penard, 1922 from a sample of freshwater Lake Agawam in the Village of Southampton, New York USA. The cells measure from 32-40 um in length. Imaged in Nomarski DIC on Olympus BH2S using SPlan 100 1.25 oil immersion objective plus variable phone camera cropping on Samsung Galaxy S24.
There are two species in this microthoracid genus: the type species T. opaca and T. australis. The cells have a dorsoventrally compressed rounded oblong body with peculiar ridging on left and right sides. There is a small anterior overhang or beak associated with the indistinct cytostome where there are two adoraal membranelles. Augustin, Foissner and Adam 1987 state that the cyrtos is invisible even with interference contrast and that the macronucleus is difficult to visualize in vivo. This is the type species T. opaca since T. australis has deeper ridges. It is interesting that both Penard 1922 and Augustin, Foissner and Adam 1987 state that food vacuoles were not seen. This sample was rich in spherical small green algae and was fed with boiled wheat seed and then neglected for two weeks. This population shows many cells distended with globular refractile food vacuoles possibly representing ingested algae.
Penard's original 19922 description: "Trochiliopsis opaca gen. nov. sp. n. Body vaguely ellipsoidal, rounded behind, convex on the right side and with no or barely noticeable convexity on the left side, curved towards the left at its part anterior and ending in a hooked beak. The animal is strongly compressed laterally, and we can then distinguish a right flank, divided into four or five longitudinal segments or ribbons separated by deep furrows; then a divided left flank in an equal number of ribbons, but more regular, clearly arched and concaved turned towards the left side. In the furrows and on both sides arise thin and long cilia which are not very abundant; forward and under the characteristic beak, they are more tight and more vigorous, and curve downwards like a hook. Under the top of the beak, on the left flank, you can see an edge (?) or double vertical line outline, which, parallel to the edge of the animal, goes straight down to a very long distance towards the posterior part of the body. Spherical nucleus, not very bulky, central. Contractile vacuole above the nucleus. Length 35 to 40 µ" (1).
"This is a very small organism, Fig. 103. Trochiliopsis opaca whose characters almost escape all in sight, and the study of which, which still remains very incomplete, could not be pursued only with great difficulty. Although the body is very pale, very pure, and contains only very small shiny grains scattered in a cytoplasm where coarser food boluses do not seem to be found, its cuticle rigid, colorless, is entirely opalescent, of such an opalescence that the light appears to be reflected in it; it is then very difficult to distinguish anything other than the contractile vesicle, then a core always vaguely outlined; longitudinal striations, on the other hand, are more clean, straight and equal on the right side (fig. 1), arched on the left side (fig. 2), where we still see, starting from the terminal beak, a very straight edge, the meaning of which cation remains enigmatic. But what is most characteristic in this organism, these are the trichocysts, small, very indistinct rods which escape the most often in plain sight, but which, after the explosion, appear in a curious form, a relatively short filament, and terminated at its distal end by two, three, or more generally four hooked points (fig. 4), divaricate, such that approximately we will soon see them again in another Infusoria (Pseudomicrothorax) where they were able be better studied. Fig. 3 shows the Infusoria seen from above, with the furrows on its two faces. Trochiliopsis opaca was found, still quite rare, at Ariana, in the leaves dead from Swan Pond" (1).
From the redescription of Augustin, Foissner and Adam 1987 "Trochiliopsis Penard, 1922 Class Kinetofragminophora: Subclass Hypostomata: Order Nassulida: Suborder Microthoracina Genus TROCHILIOPSIS Penard, 1922. Diagnosis. Microthoracidae Wrzesniowski, 1870 with cytostome in the anterior third of body. Three preoral kineties subapically on the left body side. Somatic kineties from either side terminate near the pointed beak-like region formed by the oral structures. Apex smooth. Right-most somatic kinety of the right side interrupted. Contractile vacuole located almost centrally. Freshwater, polysaprobic" (2).
"Type-species . Trochiliopsis opaca Penard, 1922. Freshwater. In vivo circa 30-40(-50) x 17-20 um. Body outline oval, anteriorly curved slightly to the ventral side terminating in a pointed beak-like region (peak). Body strongly compressed laterally (circa 2:1) and covered in a rigid longitudinally ribbed armour. Somatic kinetics in deep, crenelated furrows, which terminate near the oral peak. Macronucleus spherical, more or less centrally located, in vivo hardly discernible. Micronucleus closely attached to the macronucleus. Contractile vacuole centrally located above macronucleus, close to the right lateral surface, diameter about 4 um; contractile vacuole pore at the end of the paroral membrane. Cytoproct slightly posterior to the contractile vacuole pore, visible as black line in dry silvered specimen (Fig. 130). Pellicle rigid, colourless, opaque. Extrusomes about 3 um, fusiform, scattered over the whole body in the ribs between the furrows, show four anchor-like processes at the distal end in the exploded phase. Probably feeds on bacteria, but no food vacuoles were found. Slow, trembling and swaying movements. Probably only two adoral membranelles, located at the oral peak. Anterior adoral membranelle most likely built up by two rows, posterior one probably by three rows of kinetosomes. Paroral membrane with 8-9 paired basal bodies (Figs 120, 122, 124). Cyrtos invisible in life even with interference contrast, but slightly impregnated with protargol silver" (2).
- Penard, E. 1922. Etudes sur les infusoires d'eau douce. Georg et Cie, Geneve
- Revision of the genera Acineria, Trimyema and Trochiliopsis (Protozoa, Ciliophora)H. Augustin, W. Foissner & H. Adam. Zoologisches Institut der Universitat Salzburg. Bulletin of the British Museum (Natural History) Zoology 52 pages 197-224 ( 24 Jun 1987 )
Photos / Sounds
What
Lagynus elegansObserver
peptolabDescription
Lagynus elegans (Engelmann, 1862) Quennerstedt, 1867 from a new sampling site: Georgica Cove, a brackish (5 ppt salinity) landward offshoot of estuary Georgica Pond. Imaged in Nomarski DIC on Olympus BH2S using SPlan 40 0.70 objective plus variable phone camera cropping on Samsung Galaxy S24.
The cells measure 130-160 um in length and exhibit the classic amphora-shaped body with a flexible neck region showing four annular constrictions atop which is the oral bulge with a prominent oral basket of nematodesmata. There is an ovoid to elliposid macronucleus and for the first time I have been able to demonstrate the micronucleus. For this observation, I compare with the most recent redescription of the species, that of Jiang et al 2023 (1). Their population was somewhat smaller than the current observation (80-125 um vs 130-160 um) but their improved diagnosis gives a range of 80-200 um based on analysis of several population reports (1).
This population is unusual in that, like Liang et al 2023 who report the presence of two micronuclei, I also observed some cells with two micronuclei. Liang et al 2023 note that Engelmann (1862); Sola et al. (1990); and Foissner et al. (1995) all report a single micronucleus. "However, only one micronucleus was recorded in all the above populations, whereas the Chinese population clearly shows two micronuclei. It is noteworthy that the size difference of the micronucleus between the two populations observed by Penard (1922) was signifcant although there was only one micronucleus per cell in each population. In addition, a variable number of micronuclei was also reported within the Chinese population of the prostomatean Pelagothrix plancticola Foissner et al., 1999 (Jiang et al. 2022). Thus, the number of micronuclei may be variable within Lagynus elegans, and could be a population-level diference" (1).
"This species was originally reported by Engelmann (1862) and was subsequently redescribed several times (Foissner et al. 1995; Kahl 1930; Liebmann 1962; Penard 1922; Quennerstedt 1867; Sola et al. 1990; Wetzel 1928). Based on new data from the Chinese population, it is redefined as follows. Improved diagnosis Cell size 80–200 × 30–60 μm in vivo; amphora-shaped; single globular to long-ellipsoidal macronucleus and one or two micronuclei; rod-shaped extrusomes, irregularly distributed beneath pellicle; contractile vacuole terminally located; three to six cervical kineties and 26–50 somatic kineties. Freshwater and brackish water habitat" (1).
"Description Cell size in vivo about 80–125×30–55 μm, 100 × 30 μm on average (n=25), with a length to width ratio of 3–4:1 (Fig. 5A, C, E, F, H). Cell generally amphora-shaped, with an enlarged ellipsoidal main part, posterior end broadly rounded; starved individuals cylindroid, with posterior half of cell fattened and transparent (Fig. 5A, C, E, F, H). Neck-like region fexible and contractile, with four deep furrows (Fig. 5A, E, H). Oral bulge prominently projected from anterior end of cell, forming an apical plate; cytostome apical and circular; oral basket inverted trapezoidal, size about 8–11×4–6 μm after protargol staining; rod-shaped nematodesmata about 7 μm long in vivo (Fig. 5A, E, K, L, O). Cell surface with longitudinal ridges throughout cell length (Fig. 5G). Cytoplasm opaque and grayish due to being filled with multitudinous food vacuoles and light-refracting granules (Fig. 5A, F, H). Extrusomes 6–10 μm long in vivo, rod-shaped, irregularly distributed beneath pellicle (Fig. 5C). One macronucleus, globular to long-ellipsoidal, located in mid-cell, size about 18–33×8–17 μm after staining; two globular to ellipsoidal micronuclei, closely associated with macronucleus, size about 1–2 × 3–4 μm after DAPI staining (Fig. 5I, J). Single contractile vacuole, terminally located, about 11 μm in diameter (Fig. 5A, H). Locomotion by swimming moderately fast, with no obvious pattern" (1).
"Somatic cilia about 10 μm long in vivo; in total, 26–37 somatic kineties consisting of monokinetids (Fig. 5B, N). One dikinetidal circumoral kinety encircling anterior end of cell (Fig. 5B, D, O). Brosse region composed of three rows of kineties, each of which contains two or three basal bodies (Fig. 5B, D, M). Invariably three perioral kineties, each ring-like and composed of dikinetids (Fig. 5B, D, M, O). Five or six cervical kineties around neck-like region, arranged in rings (Fig. 5B, N)" (1).
- Morphological and molecular examination of the ciliate family Lagynusidae (Protista, Ciliophora, Prostomatea) with descriptions of two new genera and two new species from China. Limin Jiang, Congcong Wang, Saleh A. Al‑Farraj, Hunter N. Hines and Xiaozhong Hu. Marine Life Science & Technology (2023) 5:178–195 https://doi.org/10.1007/s42995-023-00174-1
Photos / Sounds
What
Order PleuronematidaObserver
davidfbirdDescription
Undulating membrane almost as long as the body. It terminates in posterior third of body in a pit mouth. Uniform ciliation with apparently 1 caudal cilium. Contractile vacuole in posterior third of body. More-or-less central ovoid nucleus. Length 65-69 µm. I think it was that long at least - I didn't record the magnification and had to rely on detective work, so there remains some small chance that it was 25-27 µm. That happens. Luckily there do not appear to be any Pleuronema that small.
Photos / Sounds
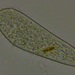
What
Paramecium (Genus Paramecium)Observer
erin34640Description
Paramecium with obvious oral groove and cilia. Moving around a larger piece of algae. Appeared to having a gliding and flipping motion. Observed in pond water sample at 10x.
Photos / Sounds
Observer
peptolabDescription
Dexiotricha granulosa Kent 1881 from the freshwater glacial kettle hole pond Kellis Pond which appeared after feeding the culture with boiled wheat seed. Imaged in Nomarski DIC on Olympus BH2 using SPlan 100 1.25 oil objective plus variable phone camera cropping on Samsung Galaxy S24. The ciliate measures um in length. The cell is elongate ellipsoid with a small subapical cytostome with small undulating membrane, two to five anterior transverse ciliary rows (paratenes), spherical macronucleus located just posterior to the equator and an adjacent central contractile vacuole with collecting vacuoles, and a caudal cilium. The most striking feature is the presence of numerous ring-shaped granules which are only seen in D. granulosa among others in the genus.
Bruce Taylor writes: "Dexiotricha granulosa is a scuticociliate, 50-70 μm long, whose cytoplasm is packed with tiny ring-shaped glyocogen granules. The cell has a distinctive pattern of kinetids at the anterior (which looks to me, like the winding path around a tiny mountain). These are not true ciliary rows or kineties but "paratenes", which appear as transverse rows running at right angles to the longitudinal kineties (see Denis Lynn, The Ciliated Protozoa, 2008, p. 43). Contractile vacuole and macronucleus are close together, at the midpoint of the cell".
Foissner et al 1994 (1) provide the differential diagnostic features: Size in vivo 40-80 x 15-30 pm, usually 50-60 um long. Shape slender to broadly ovoid and light at the mouth sunken; however, the front end is slightly truncated at an angle Macronucleus spherical approximately in the middle of the body. 1 spherical micronucleus Contractile vacuole approximately in the middle of the body. Resting extrusomes (trichocysts) rod-shaped, inconspicuous. Cytoplasm usually densely filled with approximately 2 um large, ring-shaped, highly refractive granules, which allows the cell to shine; at low magnification is dark to black. 30-38 longitudinal ciliary rows. At the rear there is a rigid caudal cilium about 28 um long. Oral apparatus subapical, small and therefore difficult to detect in vivo. 3 small adoral membranelles, to the right of it a semicircular undulating membrane; more detailed structure only visible after silver impregnation, not necessary for determination. Movement unremarkable, often standing almost still. (1)
I had previously identified a somewhat unusual population of this species in Kellis Pond sample also fed with boiled wheat seed. In that population, the cytostome was located more posteriorly at the end of the anterior 1/3 of the cell and was much larger and easy to discern with a large undulating membrane (see https://www.inaturalist.org/observations/207571468). As both Foissner and also Kreutz (realmicrolife.com) point out, the mouth of D. granulosa is subapical and small and difficult to detect in vivo (see the references and https://www.inaturalist.org/observations/207571468 for text of detailed descriptions. The current September 2024 population shows the usual cytostomal appearance of D. granulosa (depicted by Foissner et al 1992).
In trying to explain the unusual oral apparatus of the April Kellis Pond cf. Dexiotricha granulosa population, Bruce Taylor writes: "In the current state of knowledge, I suppose it qualifies as D. granulosa. However, there's no particular reason to be sure that there is only one species in nature that possesses these glycogen granules. Your population certainly has a distinctive appearance....and a fairly small size, evidently. So, it could well be something new. The term "macrostome" is usually applied to particular life stages in polymorphic species like Tetrahymena vorax. In this case, we don't know whether this is a poylmorphism or a stable trait in a local population. We don't have detailed information about ciliature etc., and lack genetic data, and have no information at all about the life cycle. Provisionally, I'd regard it as D. cf. granulosa....and investigate it more deeply, if you have time, inclination and access to a good big bloom. :)" .
So, with the finding of another population from the same site sampled 5 months later, we see a reversion to the usual cytostomal appearance of Dexiotricha granulosa. Without molecular genetic comparison of the two populations, it is impossible to discern which of Bruce's proposals: polymorphism or a stable trait in a local population, is happening here. However, with the finding of a "normal" appearing population at the same site, it seems plausible that the April 2024 population represents a seasonal, nutritional, or some other environmental polymophism in the Kellis Pond population.
- Foissner W., Berger H., Kohmann F. (1994) Taxonomische und ökologische Revision der Ciliaten des Saprobiensystems – Band III: Hymenostomata, Prostomatida, Nassulida. Informationsber. Bayer. Landesamtes Wasserwirtsch. Heft 1/94: 240-44.
- Redescription of Dexiotricha colpidiopsis (Kahl, 1926) Jankowski, 1964 (Ciliophora, Oligohymenophorea) from a Hot Spring in Iceland with Identification Key for Dexiotricha species. Zhishuai QU, René GROBEN, Viggó MARTEINSSON, Sabine AGATHA, Sabine FILKER, Thorsten STOECK. Acta Protozool. (2018) 57: 95–106
- Morphological reports on two species of Dexiotricha (Ciliophora, Scuticociliatia), with a note on the phylogenetic position of the genus Xinpeng Fan, Saleh A. Al-Farraj, Feng Gao and Fukang Gu. International Journal of Systematic and Evolutionary Microbiology (2014), 64, 680–688
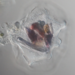
Photos / Sounds
What
Family NassulidaeObserver
someplantDescription
Magnification of all photos: 400×
Habitat: water squeezed from Spirogyra growing along the edge of a lake.
Using stage micrometer for calibration, estimated to be about 58 µm long and 38 µm wide.
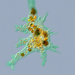
Photos / Sounds
Observer
someplantDescription
Magnification of all photos: 400×
Habitat: muddy water from the edge of a shallow pond. Associated plants: Boltonia asteroides
Using stage micrometer for calibration, estimated to be about 245 µm long and 72 µm wide.
Stats
- 8132 IDs made for others